 Granted, the plug will work, but will wear prematurely and not perform as well.Īdvice, how-to guides, and car care information featured on and AutoZone Advice & How-To’s are presented as helpful resources for general maintenance and automotive repairs from a general perspective only and should be used at your own risk. You don’t want to install single platinum plugs if your owner’s manual recommends a double platinum spark plug. This created a plug that is very durable in DIS systems, lasting upwards of 80-100K miles. What this means is that instead of the center electrode wearing as it does in traditional ignition systems, now, the side electrode wears on the plug that is reversed-polarity! This created the need for a double-platinum plug that has a platinum disc on both the center electrode, and the side electrode. On the other plug, the spark travels from the side electrode to the center electrode. This means that the spark on one plug travels from the center electrode to the side electrode. To make this even more complicated, one spark plug attached to each coil is positive polarity, while one is reverse polarity. In doing this, both spark plugs that attach to that single coil pack spark at the exact same time – the difference is that one is firing on the compression stroke, and one is firing on the exhaust stroke of the sister-cylinder, which is referred to as a “wasted spark”. 
It is thought that the total amount of iridium in the planet Earth is much higher than that observed in crustal rocks, but as with other platinum group metals, the high density and tendency of iridium to bond with iron caused most iridium to descend below the crust when the planet was young and still molten.In the early 1990’s, DIS “wasted spark” ignition systems were created, which essentially means that one coil pack controls the firing of two spark plugs in the system. Iridium is found in meteorites with an abundance much higher than its average abundance in the Earth's crust. The unusually high abundance of iridium in the clay layer at the K–T geologic boundary gave rise to the Alvarez hypothesis that the impact of a massive extraterrestrial object caused the extinction of dinosaurs and many other species 65 million years ago. 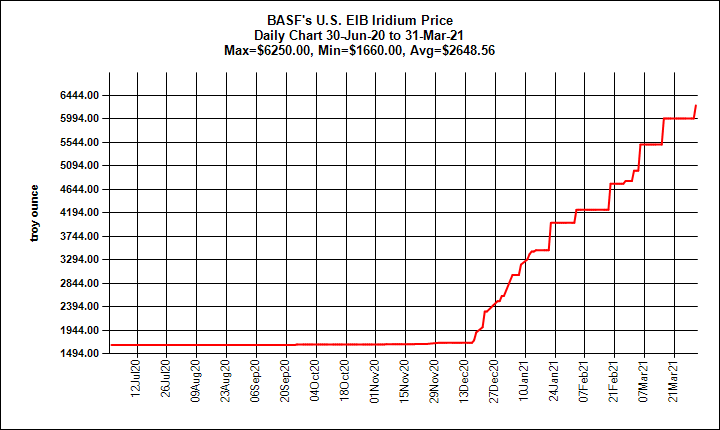
Iridium radioisotopes are used in some radioisotope thermoelectric generators. Iridium metal is employed when high corrosion resistance at high temperatures is needed, as in high-end spark plugs, crucibles for recrystallization of semiconductors at high temperatures, and electrodes for the production of chlorine in the chloralkali process. The most important iridium compounds in use are the salts and acids it forms with chlorine, though iridium also forms a number of organometallic compounds used in industrial catalysis, and in research. 191Ir and 193Ir are the only two naturally occurring isotopes of iridium as well as the only stable isotopes the latter is the more abundant of the two. Iridium is one of the rarest elements in the Earth's crust, with annual production and consumption of only three tonnes. Smithson Tennant, the primary discoverer, named the iridium for the goddess Iris, personification of the rainbow, because of the striking and diverse colors of its salts. Iridium was discovered in 1803 among insoluble impurities in natural platinum. Although only certain molten salts and halogens are corrosive to solid iridium, finely divided iridium dust is much more reactive and can be flammable. A very hard, brittle, silvery-white transition metal of the platinum family, iridium is the second-densest element (after osmium) and is the most corrosion-resistant metal, even at temperatures as high as 2000 ☌.  
Iridium ( /ɨˈrɪdiəm/ i- rid-ee-əm) is the chemical element with atomic number 77, and is represented by the symbol Ir. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
